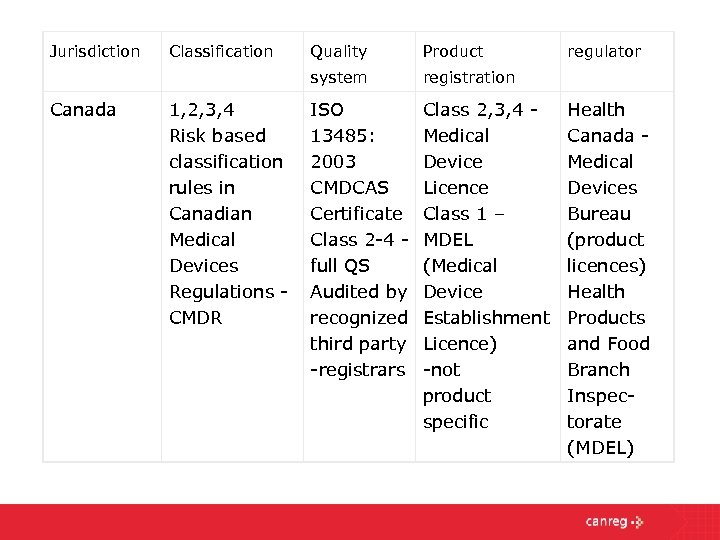
ISO 13485 is now considered to be inline standard and requirement for medical devices even with "Global Harmonization Task Force Guidelines" (GHTF). This standard adopted by CEN as EN ISO 13485:2003/AC:2007 is harmonized with respect to the European medical device directives 93/42/EEC, 90/385/EEC and 98/79/EC. The result of a positive assessment is the certificate of conformity allowing the CE mark and the permission to sell the medical device in the European Union. The preferred method to prove conformity is the certification of the Quality Management System according ISO 9001 and/or ISO 13485 and ISO 14971 by a Notified Body. The conformity of Medical Devices and In-vitro Diagnostic Medical Device according to EEC-decrees 93/42/EEC, 90/385/EEC and 98/79/EEC must be assessed before sale is permitted.
Iso 13485 registrars verification#
Specific requirements for verification of the effectiveness of corrective and preventive actions.Ĭompliance with ISO 13485 is often seen as the first step in achieving compliance with European regulatory requirements.Specific requirements for documentation and validation of processes for sterile medical devices.Specific requirements for inspection and traceability for implantable devices.Focus on risk management activities and design control activities during product development.Controls in the work environment to ensure product safety.


Quality Management System for Medical Devices - ISO 13485 : 2003 Certification
